FactoryTalk PharmaSuite — Electronic Batch Recording (eBR)
Compliant electronic batch record system — Replacing paper records for paperless production management, compliant with FDA 21 CFR Part 11 and GMP regulations
PharmaSuite eBR (Electronic Batch Record) is Rockwell Automation's electronic batch record system designed specifically for the pharmaceutical industry. It replaces traditional paper-based batch records to achieve comprehensive digital production management. The system ensures that all production operations, material usage, parameter settings, and quality data are completely recorded electronically, complying with FDA 21 CFR Part 11 electronic signatures, EU Annex 11, and GMP international regulatory requirements.
PharmaSuite eBR seamlessly integrates with existing ERP, LIMS, DCS/SCADA systems, building a complete digital production management workflow from recipe management and production execution to batch release, significantly enhancing data integrity, production efficiency, and regulatory compliance capabilities.
Core Features
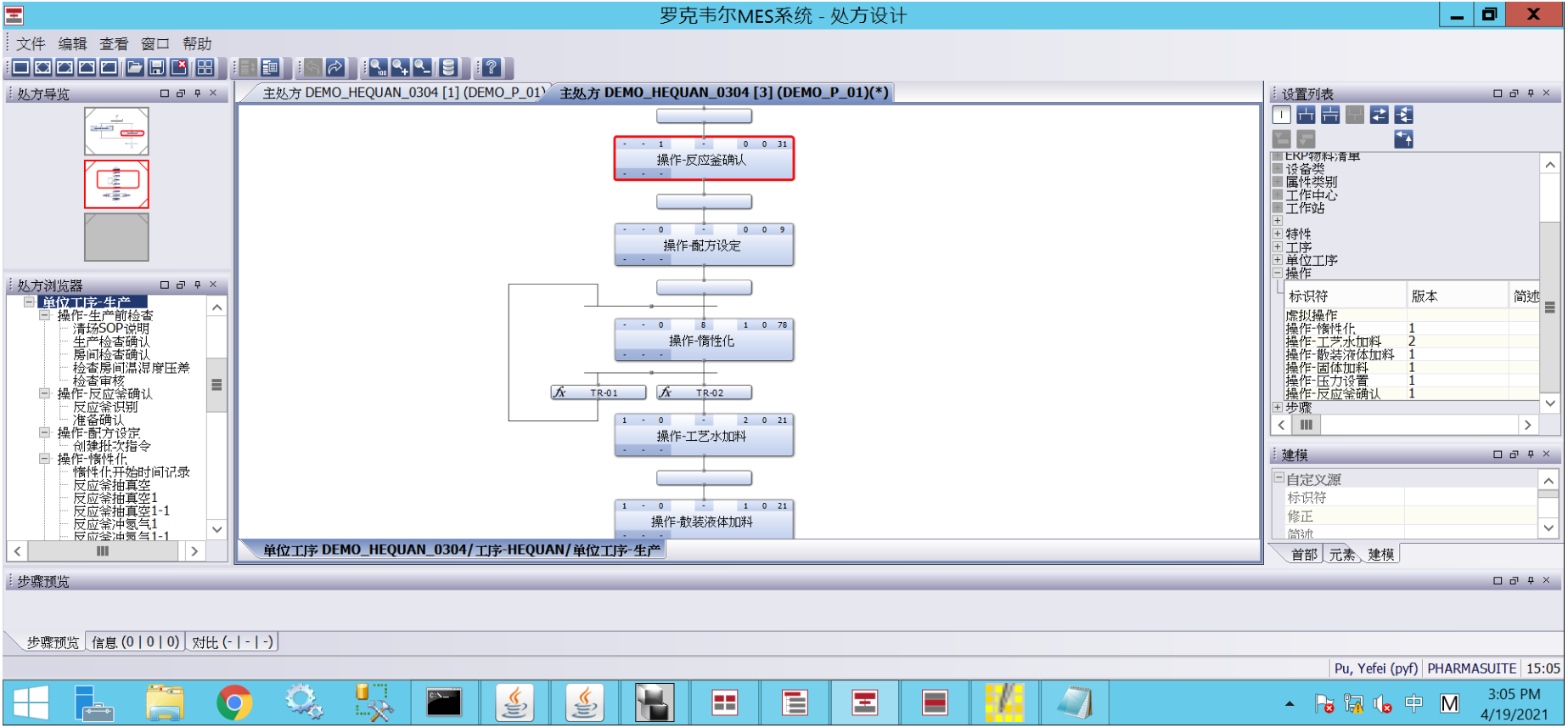
Electronic Batch Record Management
Comprehensively records all operational steps, material usage, and parameter settings for production batches, ensuring traceability and data integrity throughout the batch production process
Regulatory Compliance Assurance
Compliant with FDA 21 CFR Part 11 electronic signatures, EU Annex 11, and other international regulations, ensuring the legal validity and compliance of electronic records
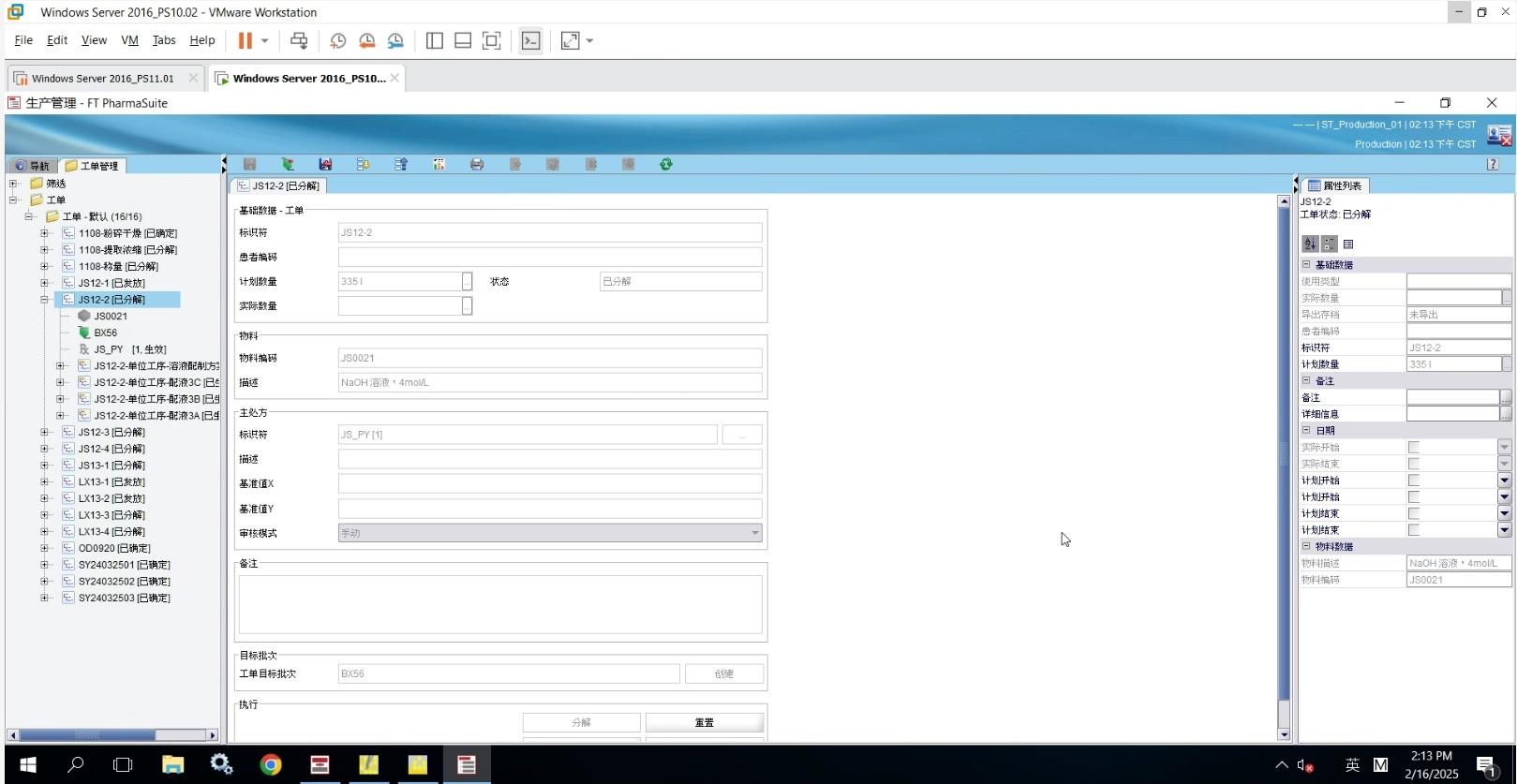
Recipe and Production Management
Supports recipe management, material management, equipment management, and quality control, achieving standardized and digital control of the entire production process
Real-Time Production Monitoring
Real-time tracking of production progress, deviation management, and exception handling, ensuring transparency and real-time control of the production process
Integration Capabilities
Seamless integration with ERP, LIMS, DCS/SCADA systems, breaking down production data silos and achieving unified information flow management
Reporting and Audit Trail
Complete audit trail, electronic signature records, and automated report generation, meeting regulatory audit and quality management requirements
Application Scenarios
Solid Dosage Production
Batch production record management for tablets, capsules, and other solid dosage forms, covering the entire process from dispensing, granulation, compression, to coating
Liquid / Injectable Production
Aseptic production batch management for liquid dosage forms and injectables, ensuring complete recording of process parameters and environmental conditions
Biopharmaceutical Production
Batch record management for vaccines, antibodies, and other biologic drugs, supporting complex bioprocess control and quality management
Traditional Medicine Production
Production batch management for herbal preparations, traditional medicine formulations, and herbal injectables, meeting the specialized recording requirements of traditional medicine manufacturing
Value Delivered
Eliminate Paper Record Errors
Reduce manual transcription errors, enhance data integrity, and ensure the accuracy and reliability of every production data point
Accelerate Batch Release
Automated review processes significantly reduce batch release time, accelerating time-to-market for products
Regulatory Compliance Assurance
Ensure all process records comply with FDA, EMA, and other international regulatory requirements, reducing audit risk
Improve Production Efficiency
Real-time monitoring and deviation management reduce downtime, improving Overall Equipment Effectiveness (OEE) and production line utilization
Related Products
Learn More About Rockwell Automation Solutions
Contact VtR Inc. for the best digital transformation solutions for your pharmaceutical operations
Contact Us