Cortellis Clinical Trials Intelligence
Clinical Trials Intelligence Platform — End-to-end workflow support from protocol design to site selection
Cortellis Clinical Trials Intelligence (CTI) is Clarivate's clinical trial planning and competitive intelligence platform. It integrates expert-curated clinical trial protocol and design information, global site data, biomarker analytics, and intuitive analysis tools into a single solution, helping pharmaceutical and biotech companies position their trials for success from day one.
The platform combines data from 35+ trial registries, peer-reviewed literature, conference abstracts, press releases, and regulatory documents, all curated by PhD- and MD-level research professionals with 25+ years of experience. Used by 85% of the world's top 20 pharmaceutical companies, serving approximately 3,000 life sciences clients worldwide.
Cortellis Clinical Trials Intelligence — Position your trial for success from day one, covering 384K+ global clinical trials
Data Coverage
Global Clinical Trials
Covering all phases, from 35+ trial registries (9 automated + 16+ manually curated)
Clinical Trial Sites
Across 200+ countries, searchable by therapeutic area, performance metrics, and geography
Biomarkers Tagged to Trials
Tagged to 302,000+ trials with unparalleled depth
Diseases Covered
Across 30+ therapeutic areas, including 34,000+ rare disease-related trials
Trial Sponsors
Global trial sponsor tracking and competitive monitoring
Literature Citations
From the Derwent Drug File (since 1964), supplemented by 84,000+ conference reports and 425,000+ press releases
Core Features
Protocol Design Support
Industry analyst-curated clinical trial protocol data, analyzing inclusion/exclusion criteria, trial arms, and endpoints. Benchmark protocols against competitors by disease area and mechanism of action to reduce avoidable protocol amendments (median cost of $535,000 per Phase III amendment)
Site Selection Analytics
277,000+ sites across 200+ countries. Integrated epidemiology data providing country-level incidence/prevalence for 1,200+ diseases. U.S. claims data overlay: 6,000+ hospitals, 2.4 million+ practitioners, 5,400+ health systems
Biomarker Analytics
813,000+ biomarkers tagged to 302,000+ trials. Discover competitor-specific biomarker application strategies and support biomarker identification and selection in protocol design
Endpoint Analysis
Curated endpoint data across all trials, benchmark trials by endpoint and timeline. Analyze trial success or failure based on endpoint outcomes with historical endpoint benchmarking
Competitive Trial Landscape
Track competitive trial strategies by disease area, patient segment, design, and mechanism of action. Trial timeline viewer provides visual charts, monitoring 46,000+ trial sponsors
Enrollment Analytics
Site-level enrollment forecasting using built-in analytics tools, tracking enrollment duration and rates. Drill down from therapeutic area to trial count to country to enrollment timeline
Rare Disease & Orphan Drug Intelligence
34,000+ rare disease trials and 3,300+ orphan drug designations. Exclusive rare disease coverage not available from competitive platforms, with site identification for niche patient populations
Cross-Module Integration
Seamless SSO connection with all Cortellis suite products, one-click navigation to pipeline (CCI), deals (CDI), drug discovery (CDDI), and regulatory intelligence
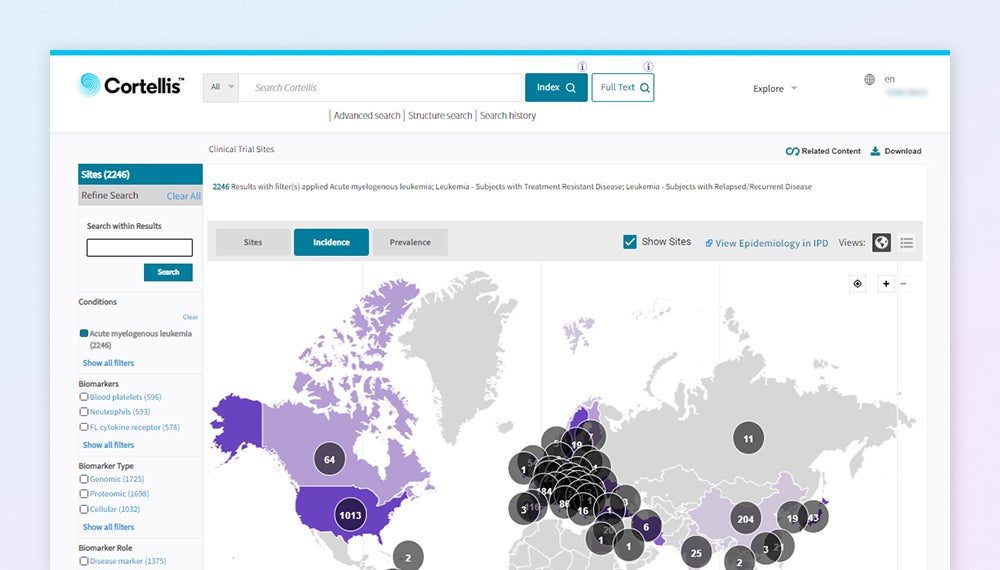
CTI Site Selection — Global trial site distribution map with integrated epidemiology data and biomarker filtering
Therapeutic Area Coverage
Complete clinical trial landscape across 30+ therapeutic areas:
Oncology
Solid tumors, hematologic malignancies
CNS
Neurology-related trial landscape
Immunology
Autoimmune disease trials
Rare Diseases
Orphan drug designations — a key differentiator
Infectious Diseases
Global infectious disease trial monitoring
Cardiovascular
Cardiovascular trial benchmarking
Respiratory
Respiratory disease trial tracking
Medical Devices
89,000+ medical device trials specifically indexed
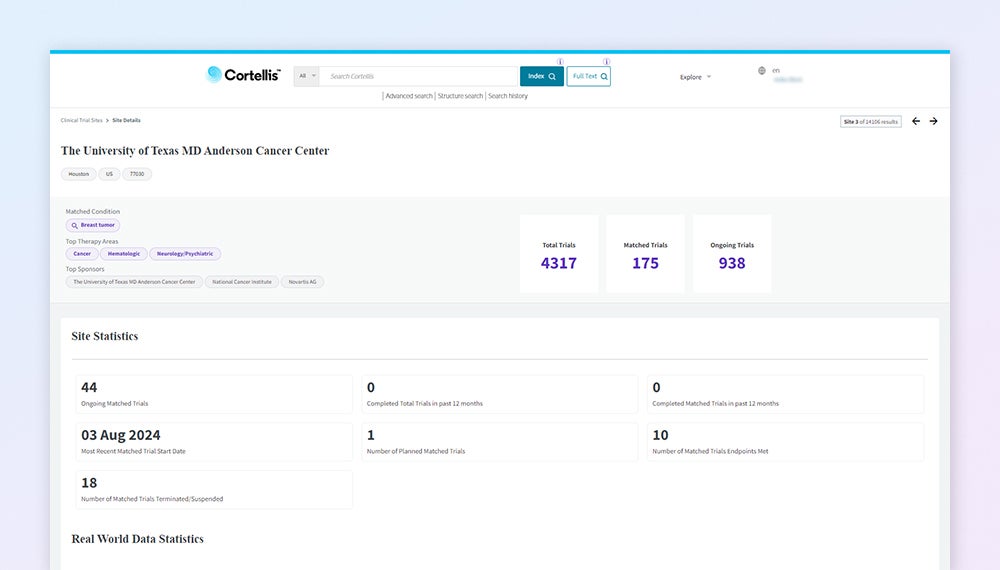
CTI Trial Feasibility Analysis — MD Anderson Cancer Center trial site statistics and performance metrics
Industry Validation
Top 20 Global Pharma Companies
Use the Cortellis platform to support clinical trial decisions
U.S. NME-Submitting Companies
Supported by Cortellis intelligence
Breakthrough Therapy Designations
Companies receiving breakthrough therapy designations use Cortellis
Prediction Accuracy
Approximately 4x more accurate in predicting Phase I success rates than traditional methods (DTSR)
Industries & Users
| Industry | Primary Use Cases |
|---|---|
| Pharmaceutical Companies | Protocol design, site selection, competitive trial monitoring, enrollment planning |
| Biotech Companies | Rare disease trial planning, biomarker strategy, enrollment feasibility |
| Medical Device Companies | Device trial intelligence (89,000+ device trials), site selection |
| Contract Research Organizations | Independent data validation, site performance benchmarking |
| Academic Medical Centers | Clinical research planning, educational use, trial landscape analysis |
| Life Sciences Investors | Clinical-stage asset evaluation, trial success assessment |
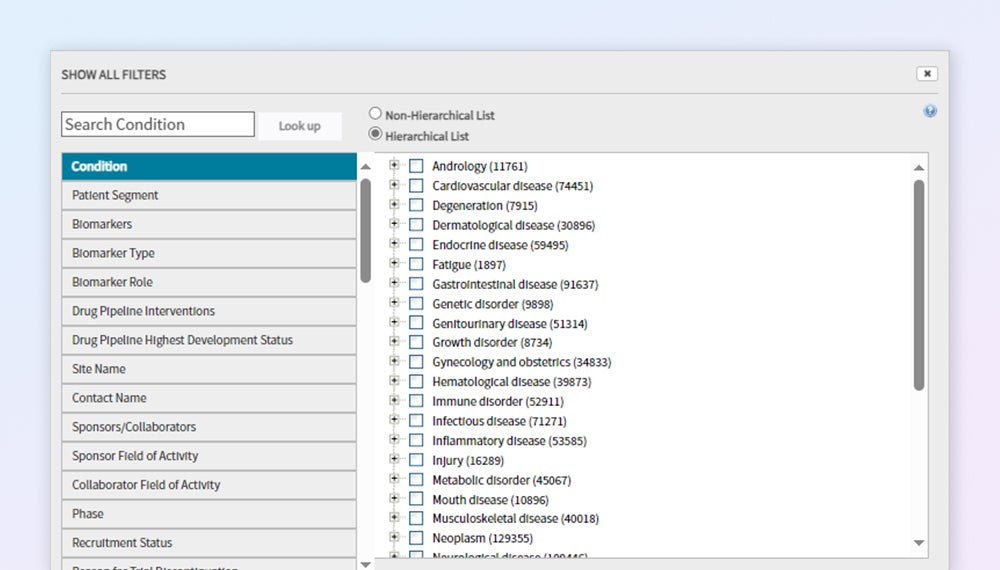
CTI Protocol Design Tool — Filter and benchmark trials by disease, patient segment, biomarker, and more
Related Products
Learn About Clinical Trials Intelligence
From protocol design to site selection, position your clinical trial for success
Contact Us