eCTD247
Cloud SaaS Electronic Submission Platform — eCTD 4.0 Pioneer, Trusted by 450+ Life Science Clients Worldwide
eCTD247 is Ennov's cloud-based SaaS platform for authoring, managing, validating, and tracking eCTD (electronic Common Technical Document) regulatory submissions. Positioned as "simple, fast, and affordable," eCTD247 enables pharmaceutical and biotech companies to assemble and submit regulatory dossiers without the complexity and cost of traditional enterprise-grade publishing systems.
Ennov is a pioneer in eCTD 4.0 — one of only four technology vendors worldwide to have successfully submitted eCTD 4.0 test sequences to the EMA, completing 11 successful eCTD 4.0 pilot submissions with EMA, FDA, and PMDA. The company serves 450+ life science clients with 500,000+ users globally.

Global eCTD Regulatory Updates — covering eCTD version upgrades, regional specification differences, and implementation timelines
Ennov Regulatory Publishing Product Portfolio
eCTD247 is positioned as the entry-level solution in Ennov's three-tier regulatory publishing product portfolio:
| Product | Type | Target Audience |
|---|---|---|
| eCTD247 | Cloud SaaS | Small to mid-sized pharma, biotech startups, companies with lower submission volumes |
| Ennov Dossier | Enterprise Publishing (Ennov native) | Mid to large pharma requiring complete dossier lifecycle management |
| InSight Publishing | Enterprise Publishing (acquired from Calyx/Liquent) | Large/top-5 pharma, complex global submissions |
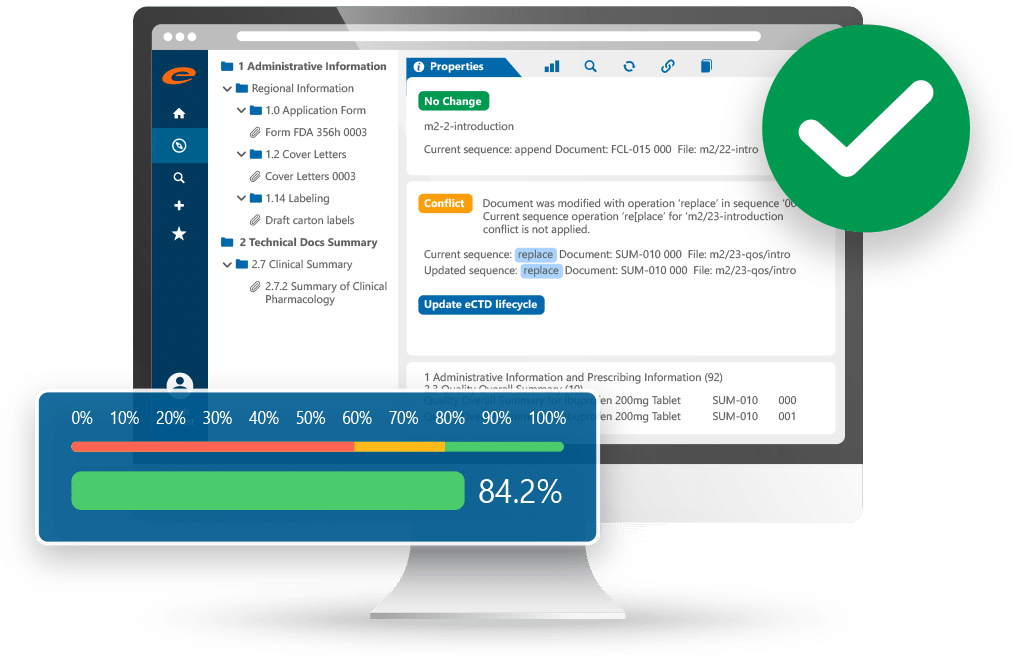
eCTD247 Submission Management — eCTD document tree structure, lifecycle management, and validation progress tracking
Core Features
eCTD Submission Authoring & Publishing
Author, publish, validate, and submit IND, NDA, ANDA, BLA, and MAA applications. Supports eCTD, NeeS, VNeeS, CTD (paper), eCopy formats, with STF generation and automatic cross-reference calculation
Document Management (Integrated EDMS)
Metadata-based architecture for rapid document retrieval, configurable database views and search engine, document validation workflows, check-in/check-out coordination, and full version management
Validation & Quality Assurance
Built-in EMA-approved eCTD validation engine with comprehensive interactive reports. Supports advanced hyperlink analysis, bookmark validation, STF validation, and IQ/OQ/PQ validation services
Collaboration & Security
Web-based interface accessible anytime anywhere, two-factor authentication and SSL encryption, public/private workgroup access control, synchronized review and electronic signatures, fully 21 CFR Part 11 compliant
Cloud Advantages (Premium Plan)
Automatic software updates instantly reflecting regulatory standard changes, claimed 48% cost reduction, no IT resources needed for maintenance, data hosted in US or EU, includes 30-day free trial
eCTD 4.0 Readiness
One of only four vendors to have successfully submitted eCTD 4.0 test sequences to EMA, with 11 pilot submissions (8 EMA + 3 FDA), collaborating with top pharma on PMDA eCTD 4.0 pilots
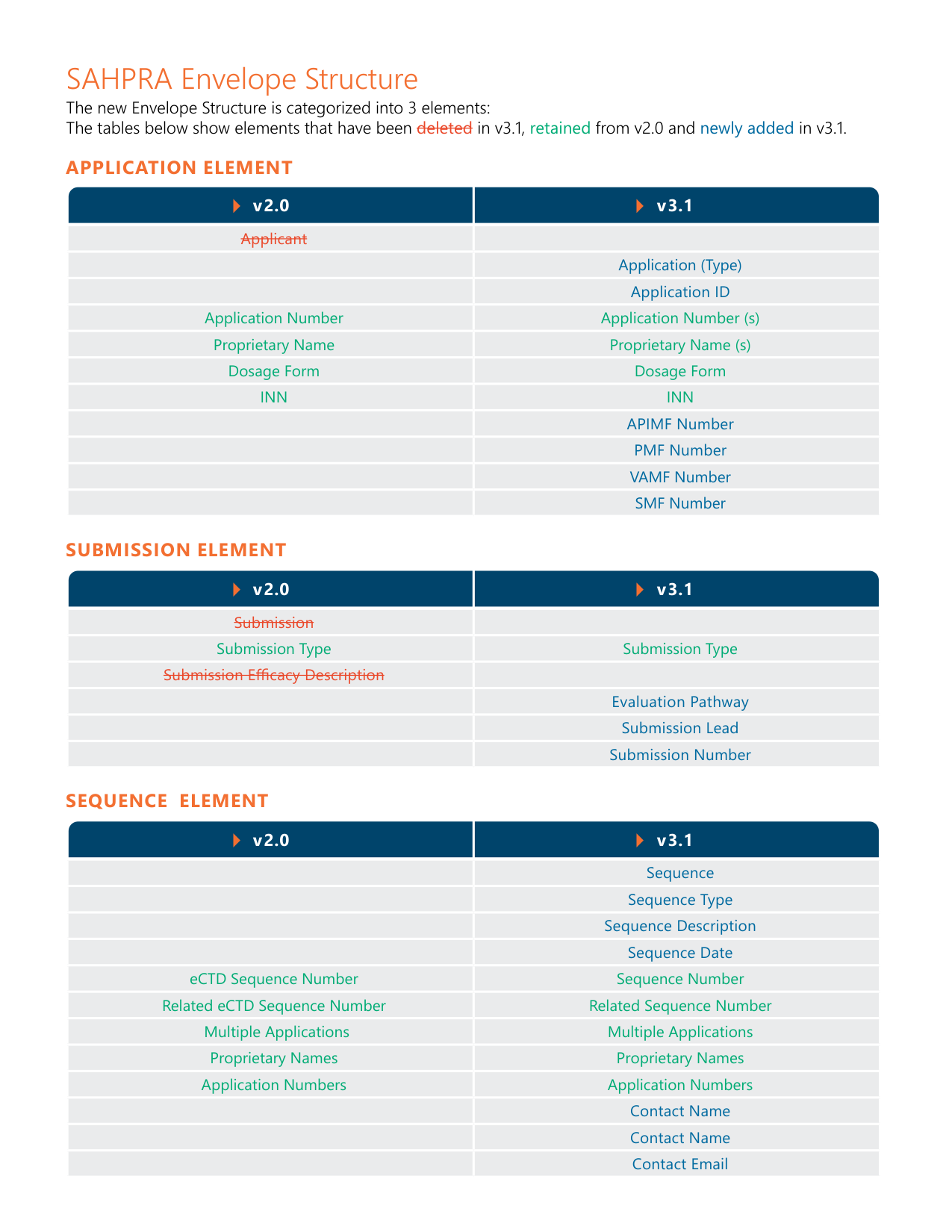
SAHPRA eCTD v2.0 vs. v3.1 Structure Comparison — color-coded highlighting of added, retained, and removed elements
Technical Specifications
| Specification | Details |
|---|---|
| Architecture (Premium) | 100% Cloud SaaS |
| Architecture (Enterprise) | On-premise deployment on customer infrastructure |
| Data Hosting | US or EU data centers (customer's choice) |
| eCTD Versions | eCTD v3.x (current), eCTD v4.0 (via InSight Publishing) |
| Supported Formats | eCTD, CTD (paper), NeeS, VNeeS, eCopy (including 510(k), PMA) |
| Submission Types | IND, NDA, ANDA, BLA, MAA |
| Validator | Built-in EMA-approved eCTD validation engine |
| Compliance | 21 CFR Part 11, ISO 9001:2015 |
| Security | Two-factor authentication, SSL encryption, access control |
| EDMS | Integrated Ennov Doc EDM (Premium plan) |
| DIA Reference Model | Pre-configured to align with the DIA EDM Reference Model |
Supported Regions & eCTD 4.0 Timeline
| Region / Authority | Current Status | eCTD 4.0 Timeline |
|---|---|---|
| FDA (USA) | eCTD mandatory for NDA/BLA/ANDA/IND | v4.0 voluntary from Sep 2024; mandatory ~2029 |
| EMA (EU) | eCTD mandatory for all centralized procedures | v4.0 optional from Dec 2025; mandatory 2028 |
| PMDA (Japan) | eCTD accepted | v4.0 mandatory from Apr 2026 |
| Health Canada | eCTD mandatory for NDS/SNDS/ANDS | v4.0 optional 2026; mandatory 2028 |
| TGA (Australia) | eCTD required for prescription medicines | v4.0 technical pilot H1 2025 |
| SAHPRA (South Africa) | Mandatory via portal since Oct 2024 | Module 1 v3.1 effective Nov 2024 |
| Swissmedic (Switzerland) | eCTD accepted | Pilot 2026; mandatory 2028 |
| GCC | eCTD accepted | Regional Module 1 specifications apply |
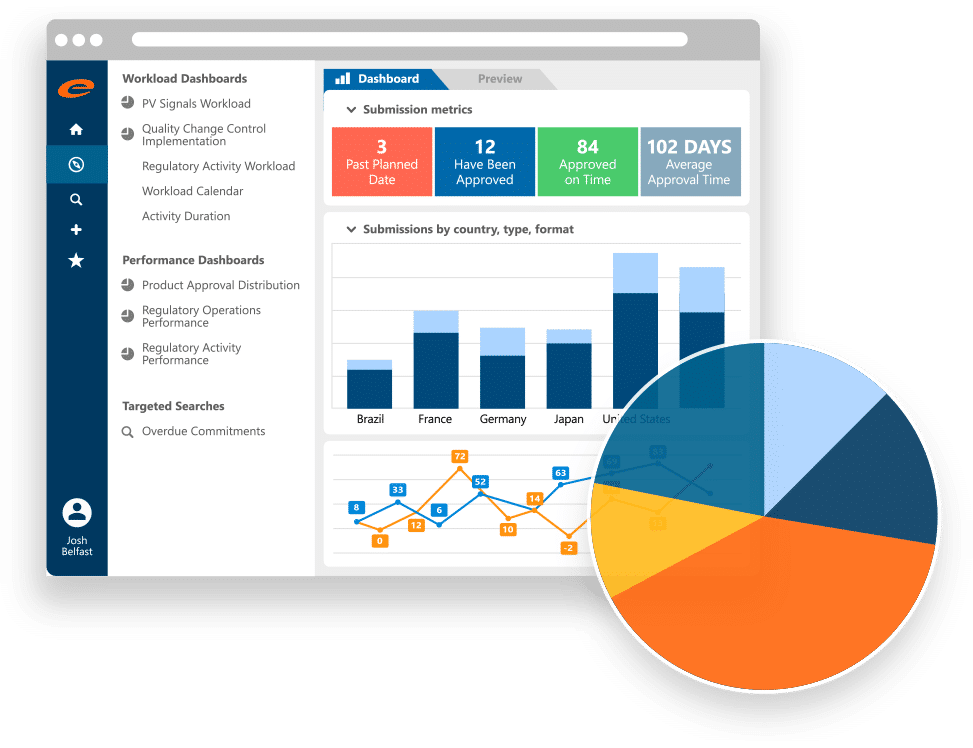
Ennov RIM — Global regulatory information management dashboard covering submission metrics, country distribution, and review timeline analysis
Application Areas
Pharmaceutical
eCTD submission authoring, dossier lifecycle management, regulatory publishing, NDA/ANDA/BLA/MAA applications.
Biotechnology
IND/BLA submissions, regulatory dossier assembly, multi-region submission management.
Generics
ANDA submissions, affordable eCTD publishing solutions.
Contract Research Organizations (CRO)
Publishing services for sponsors, multi-client submission management.
Animal Health
Veterinary NeeS (VNeeS) submissions, regulatory compliance.
Medical Devices
eCopy submissions (510(k), PMA).
Competitive Comparison
| Feature | Ennov | Lorenz docuBridge | Certara GlobalSubmit | Veeva Vault Submissions |
|---|---|---|---|---|
| Deployment | Cloud, on-premise, hybrid | On-premise or hosted | Cloud SaaS only | Cloud SaaS only |
| eCTD 4.0 | 11 pilots (EMA/FDA/PMDA) | Forward compatible | Ready | Ready |
| Integrated EDMS | Native (Ennov Doc) | External | No | Native (Vault) |
| Validator | Built-in (EMA approved) | Bundled (eValidator) | Built-in | Built-in |
| AI Capabilities | Yes (native, v11.0) | Limited | Limited | Moderate |
| Submission Legacy | 1M+ (Calyx/InSight) | Undisclosed | 1.5M+ | Undisclosed |
Key Differentiators
Three-Tier Product Portfolio
The only vendor offering self-service SaaS (eCTD247), enterprise native (Dossier), and enterprise acquired (InSight Publishing) solutions — covering startups to top-5 pharma
eCTD 4.0 Leadership
One of only 4 vendors to complete EMA pilots; 11 pilot submissions among the highest in the industry
Rapid Time to Value
eCTD247 Premium goes live "within days," with a 30-day free trial. Cloud model claims 48% cost reduction
1M+ Submission Legacy
Calyx/InSight Publishing track record of over 1 million global regulatory submissions, with deployment flexibility to switch between cloud and on-premise at any time

eCTD247 Electronic Signatures — 21 CFR Part 11 compliant document signing, distribution, and lifecycle management
Customer & Industry Validation
| Metric | Data |
|---|---|
| Life Science Clients | 450+ |
| Global Users | 500,000+ |
| Global Regulatory Submissions | 1M+ (via Calyx/InSight legacy) |
| On-Time Project Delivery | 98.5% |
| Maintenance Renewal Rate | 96% |
| Customer Audit Success Rate | 100% |
Related Products
Learn More About eCTD247
Accelerate your drug application process, ensure global regulatory compliance, and experience a 30-day free trial today
Contact Us