Certara
Global Leader in Model-Informed Drug Development (MIDD)
Integrating biosimulation software, AI technology platform and consulting services to accelerate drug development from discovery to market
Certara, Inc. is the global leader in Model-Informed Drug Development (MIDD), offering biosimulation software, technology platforms and consulting services to accelerate drug development from discovery to market. The company combines a comprehensive software platform ecosystem with over 1,100 scientists and experts to support drug development across all therapeutic areas.
Adopted by over 2,400 biopharma companies, academic institutions and regulatory agencies
Since 2014, over 90% of FDA new drug approvals have been supported by Certara software or services
Combining a comprehensive software ecosystem with top talent to support all therapeutic areas
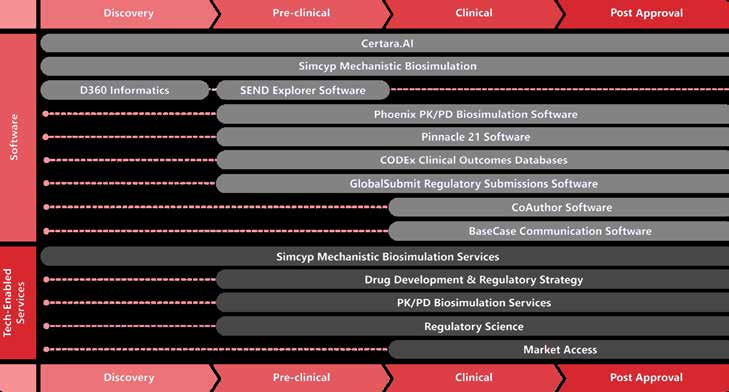
Drug Development Lifecycle Positioning
Certara's integrated software and consulting ecosystem spans the entire drug development lifecycle, providing corresponding products and services at each stage.
Drug Discovery
D360 (Scientific Informatics Platform), Secondary Intelligence (Safety Assessment), Certara.AI (Life Sciences GPT)
Preclinical
Simcyp Discovery (FIH Dose Prediction), SEND Explorer (Data Review), PBPK Modeling
Clinical (Phase I-III)
Phoenix PK/PD (NCA, PopPK, PKPD), Trial Simulator, NLME, Simcyp (DDI, Special Populations), Pinnacle 21 (CDISC), Integral (Data Management)
Regulatory Submission
Pinnacle 21 (Data Validation), GlobalSubmit (eCTD), CoAuthor (Medical Writing), MIDD Consulting, Clinical Pharmacology Consulting
Post-Market
Market Access Radar, BaseCase (Value Communication), HEOR, Real-World Evidence, Label Claim Support
Cross-Stage
Certara.AI, DIDB (Drug Interactions), CODEX (Clinical Outcomes), MBMA Consulting
Drug Discovery & Preclinical Platform
D360 — Scientific Informatics Platform
The industry-leading self-service scientific data informatics platform designed for drug discovery. Used by over 6,000 research scientists globally for small molecule and biologics discovery and preclinical safety assessment.
Multi-Modality Support
Supports small molecules, peptides, oligonucleotides, antibodies, ADCs and other modalities
Self-Service Data Query
Drag-and-drop interface with shareable queries, standardized data cleansing, unit handling, data type recognition and aggregation control
Advanced Visualization & API Integration
Scatter plots, histograms, cluster analysis; Python API wrapper and REST API for AI/ML data pipeline integration
D360 Express
Out-of-the-box integrated platform for accelerated deployment; integrates with Design Hub, Scilligence, CDD Vault, Optibrium
SEND Explorer — Preclinical Data Review
A cloud-based validated application for aggregating, visualizing and interactively interpreting SEND-format preclinical data. Designed by frontline researchers to support rapid review of nonclinical study results.
Study Data Interpretation
Group/individual study interpretation with clinical and histopathology trends over time
Multi-Study Comparison & Cloud Collaboration
Multi-study and control group comparison with cloud architecture for easy sharing and collaboration
Secondary Intelligence — Off-Target Safety Prediction
Collects, curates and visualizes all secondary pharmacology assay results, providing quantitative off-target safety risk scores for each compound. Risk levels are indicated with red/yellow/green coding for potential adverse reactions. Currently the only tool on the market addressing this translational challenge.
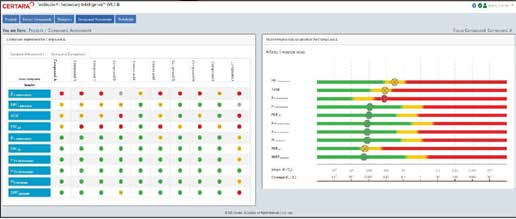
ToxStudio Integration Module
The first module of the Certara ToxStudio integrated modeling and simulation platform
Quantitative Risk Assessment
Quantifies the probability of off-target safety issues that may hinder clinical advancement
Phoenix PK/PD Platform
The gold standard for global PK/PD analysis with over 30 years of history. Used by over 6,000 researchers across 1,500+ institutions in 60 countries, including 11 divisions of the US FDA (250+ licenses), PMDA, CFDA/NMPA and MHRA. Up to 90% of FDA-approved new drugs used the Phoenix platform for critical support during development.
| Product | Description |
|---|---|
| Phoenix WinNonlin | Industry-standard noncompartmental analysis (NCA), pharmacokinetic/pharmacodynamic (PK/PD) and toxicokinetic (TK) modeling |
| Phoenix NLME | Population PK/PD (nonlinear mixed effects) modeling software with user-friendly yet powerful interface; used by US FDA for over 10 years |
| Phoenix IVIVC Toolkit | In vitro-in vivo correlation analysis tool designed for formulation scientists to improve bioequivalence study success rates |
| Trial Simulator | Optimize clinical trial design through computer-aided simulation, evaluating "what-if" scenarios for informed experimental design decisions |
| RsNLME / Pirana | R-based Certara NLME engine and NONMEM interface, enabling scientists to run population PK/PD models from the R command-line environment |
PBPK & QSP Solutions
Simcyp PBPK Simulator
The world's most widely adopted PBPK modeling and simulation platform, developed over 25 years in collaboration with the Simcyp Consortium (37 leading pharmaceutical companies). Licensed by 11 regulatory agencies worldwide.
New Drugs Approved via Simcyp in Lieu of Clinical Trials
Over 80% of FDA PBPK-related approvals between 2019-2023 used Simcyp
Drug Label Claims
Over 400 drug label claims based on Simcyp simulation results
EMA Qualification (2025)
Allows Simcyp to replace up to 6 CYP-mediated DDI clinical trials
First FDA Virtual Bioequivalence Approval
First complex generic drug approved through computer simulation in 2019
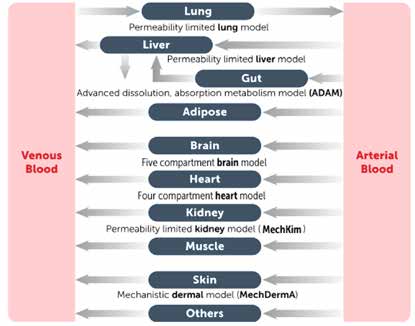
| Simcyp Product Line | Description |
|---|---|
| Simcyp Discovery | Early-stage FIH (First-in-Human) and IND application PBPK modeling |
| Simcyp Biopharmaceutics | CMC and formulation-oriented PBPK with ADAM/M-ADAM models, IVIVC, SIVA and Virtual Bioequivalence (VBE) modules |
| Simcyp Pediatric | Industry's most advanced pediatric PBPK technology, covering neonates to adolescents |
QSP Consulting Services & Certara IQ
Quantitative Systems Pharmacology (QSP) combines computational modeling with experimental data to study drug-biology-disease relationships. Certara IQ (October 2025) is an AI-driven QSP modeling platform providing validated models, standardized workflows and regulatory-compliant insights.
| Specialized Tool | Description |
|---|---|
| Immunogenicity (IG) Simulator | Predicts immunogenicity incidence and PK/PD impact |
| Vaccine Simulator | Optimal vaccine dosing strategies |
| Neuroscience QSP Models | Alzheimer's disease, Parkinson's disease, psychiatric disorder modeling |
Therapeutic Areas: Oncology, Vaccines, Neurology, CNS, Hematology, Autoimmune, Rare Diseases, Dermatology, Gene Therapy.
DIDB — Drug Interaction Database
The industry's most comprehensive drug interaction resource, originally developed by the University of Washington and trusted by over 200 pharmaceutical companies and regulatory agencies.
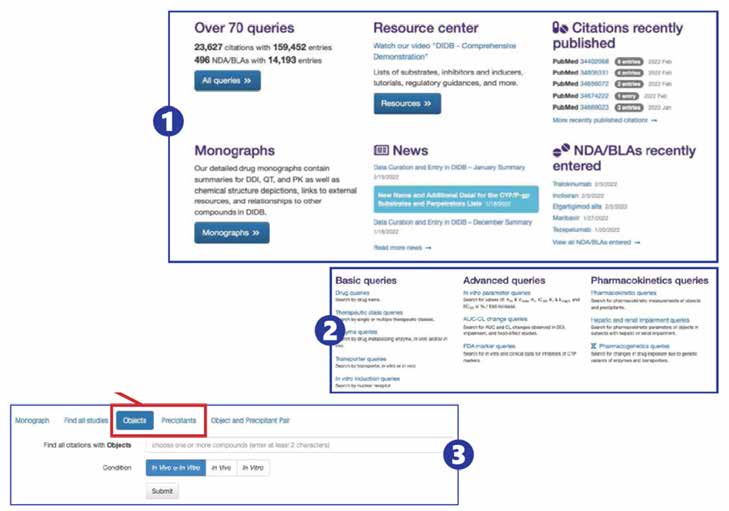
Citation Coverage
Covering publications and FDA regulatory documents, with 630+ NDA/BLA packages updated monthly
Pre-Built Queries
For data retrieval, in-depth drug monographs and DDI calculators
Comprehensive Coverage
Metabolic DDI, transporter DDI, absorption DDI, pharmacogenomics, food effects, organ impairment
CDISC Compliance & Data Standards
Pinnacle 21 Enterprise — CDISC Compliance
A clinical data standardization data science platform compliant with CDISC standards (SDTM/ADaM). Trusted by over 130 organizations, including the FDA and PMDA. 22 of the top 25 biopharma companies by R&D spending use Pinnacle 21.
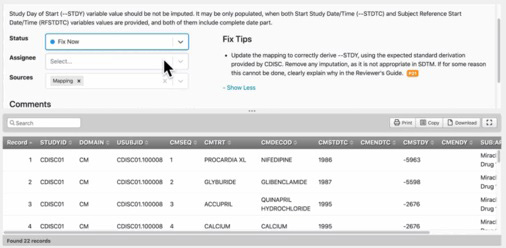
Dashboards & Scoring Algorithms
Monitor submission data readiness with change tracking across multiple validation runs
Define.xml Preparation Efficiency Improvement
Significantly reduces CDISC Define.xml document preparation time
Pinnacle 21 Enterprise Plus (September 2025)
No-code interface, 50% reduction in specification drafting time, API integration with SAS and R, less than 1 month onboarding time
Formedix ryze — Clinical Data Management & SDTM Automation
A cloud-based Clinical Metadata Repository (CMDR) and automation suite that can complete CDISC-compliant clinical study design and build in 6 weeks.
Study Design Phase Prediction
Predicts source dataset column headers during the study design phase
Automated SDTM Mapping
Pre-maps source data to CDISC SDTM standards to ensure data collection completeness
Regulatory Submission & Medical Writing
CoAuthor — AI-Powered Regulatory Writing
Generative AI-driven regulatory and medical writing software. Using a "human-in-the-loop" approach to reduce first draft production time by over 30%.
Multiple Document Types
Patient narratives, Clinical Study Reports (CSR), protocols, summaries, toxicology reports
Cloud Architecture & Integrations
Microsoft Word integration, complete eCTD template library, structured content authoring; Veeva AI Partner Program integration (October 2025)
GlobalSubmit — eCTD Submission Software
Cloud-based eCTD submission quality control and publishing tool that reduces quality control processes from hours to minutes. 21 CFR Part 11 compliant.
Comprehensive Application Type Support
IND, NDA, ANDA, sNDA, BLA, MAA, DMF, ASMF, NDS
System Integration
Integrates with major document management systems
Integral — Data Repository
A 21 CFR Part 11 compliant cloud-based data management system for clinical pharmacology, quantitative pharmacology and biostatistics data.
Regulatory Compliance
Intuitive interface with complete data traceability meeting regulatory audit requirements; validation and maintenance managed by Certara
Market Access Solutions
BaseCase — Market Access Value Communication
A no-code application builder for interactive market access tools. Supports face-to-face communication with payers and healthcare professionals through real-time calculations and data visualization.
Interactive Tools
Interactive presentations, pricing models, ROI calculators
Country Adaptation & Compliant Deployment
Translation and localization templates included to simplify legal approval processes
Market Access Radar — Market Access Intelligence
A curated pharmaceutical market access database and intelligence service with over 8 years of historical data. Updated daily by reimbursement experts.
Smart Search & Custom Data Sources
MeSH-integrated search engine, customizable by country, category and tags; covering reimbursement decisions, HTA publications, regulatory changes
Certara.AI — The AI Platform Designed for Life Sciences
A secure enterprise-grade platform for deploying life sciences-specific GPTs. Access to over 60 million life sciences research publications. Trained on biomedical data to understand scientific concepts and deliver highly specific, validated responses.
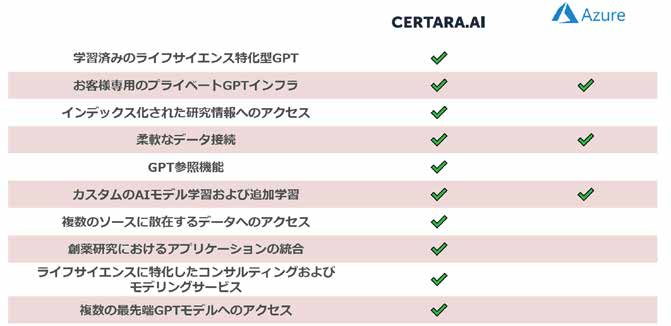
Life Sciences-Specific GPT
Trained specifically on biomedical data with real-time data indexing for the latest insights
Private Infrastructure & Flexible Architecture
Isolated, dedicated or on-premises deployment; flexible data connections to organizational internal data; AI model-agnostic architecture
Consulting Services
Over 650 experts globally covering clinical pharmacology, quantitative pharmacology, regulatory strategy and market access. Certara has completed over 8,000 projects with clients in the past decade.
| Service Area | Description |
|---|---|
| Model-Informed Drug Development (MIDD) | End-to-end MIDD strategy consulting spanning from discovery to post-market |
| Clinical Pharmacology | Gap analysis covering 40+ questions evaluated by FDA in NDA clinical pharmacology data packages; from FIH dose prediction to NDA submission support |
| FDA Project Optimus | Specialized consulting for oncology dose optimization; Certara advanced 500+ oncology programs in the past year alone |
| MBMA & CODEX | Model-based meta-analysis combined with the CODEX database covering published clinical trial outcome data across 60+ therapeutic areas |
| Regulatory Writing & Medical Publishing | 30+ years of regulatory document writing experience covering all ICH modules, all major regulatory agencies and all document types |
| Market Access, HEOR & Real-World Evidence | Decision analysis and modeling, health economics, market access and pricing strategy, real-world evidence, payer communication and value dissemination |
Key Metrics & Technical Specifications
| Item | Value |
|---|---|
| Customers | 2,400+ organizations across 66 countries |
| Scientists & Experts | 1,100+ |
| FDA New Drug Approval Support Rate | Over 90% (2014-2024) |
| Phoenix Users | 6,000+ scientists, 1,500+ institutions, 60 countries |
| Phoenix FDA Licenses | 250+ licenses across 6 centers |
| Simcyp Regulatory Agency Licenses | 11 agencies worldwide |
| New Drugs Approved via Simcyp | 120+ |
| Simcyp Label Claims | 400+ |
| Pinnacle 21 User Organizations | 130+ (including FDA, PMDA) |
| D360 Users | 6,000+ discovery research scientists |
| DIDB Citations | 27,000+ |
| DIDB Customers | 200+ pharma/biotech companies |
| Certara.AI Literature Library | 60 million+ life sciences publications |
| CoAuthor Efficiency Gain | 30%+ reduction in first draft production time |
| CODEX Therapeutic Areas | 60+ |
| Client Projects (Past Decade) | 8,000+ |
| Peer-Reviewed Publications (2024) | 100+ |
| Stanford Top 2% Scientists (2024) | 12 Certara scientists |
Target Industries
| Industry | Applications |
|---|---|
| Pharmaceuticals (Big Pharma) | End-to-end drug development from discovery to market access; 22 of the top 25 biopharma companies by R&D spending use Certara products |
| Biotech Companies | Biosimulation and regulatory strategy for emerging biotech companies with limited internal capabilities |
| Generics/Biosimilars | Virtual Bioequivalence (first FDA VBE approval in 2019), complex generics, transdermal BE (MechDermA) |
| Contract Research Organizations (CROs) | PK/PD analysis, PBPK consulting, regulatory writing, data standardization |
| Regulatory Agencies | FDA (250+ Phoenix licenses, 11 Simcyp divisions), PMDA, EMA, NMPA/CFDA, MHRA and others |
| Academia | Certara University training, academic centers of excellence, Simcyp Consortium |
| Medical Devices/IVD | Regulatory submission (510(k), PMA) and medical writing support |
Therapeutic Area Coverage
| Therapeutic Area | Key Capabilities |
|---|---|
| Oncology | Project Optimus dose optimization, 500+ programs per year, RTOR submissions, combination therapy QSP |
| Rare/Orphan Diseases | 90% of FDA orphan drug approvals since 2014; 130+ rare disease submissions; approval achieved with as few as 6 patient data points |
| CNS | Alzheimer's disease, Parkinson's disease, psychiatric disorder QSP models |
| Cardiovascular | Therapeutic modeling and concentration-QTc analysis |
| Immunology | Immunogenicity Simulator (QSP), immune-mediated disease modeling |
| Infectious Diseases | Antiviral, antibacterial, antiparasitic modeling |
| Metabolic/Endocrine | Diabetes and endocrine disease development support |
| Pediatrics | Simcyp Pediatric (neonates to adolescents), population diversity PBPK models |
| Vaccines | Vaccine Simulator, optimal dosing strategies |
| Gene Therapy | QSP consulting and biosimulation support |
| Dermatology | MechDermA model for topical drug development and virtual BE |
| Radiopharmaceuticals | Preclinical radiopharmaceutical development QSP modeling |
Key Validated Outcomes
FDA New Drug Approval Support
Over 90% of new drug approvals between 2014-2024 supported by Certara software or services
Simcyp PBPK FDA Dominance
Over 80% of FDA PBPK-related approvals between 2019-2023 used Simcyp
First FDA Virtual BE
First complex generic drug approved through computer simulation in 2019
EMA DDI Qualification (2025)
Simcyp can replace up to 6 CYP-mediated DDI clinical trials
Drug Label Claims
400+ label claims based on Simcyp simulation results; 100+ new drugs approved via Simcyp in lieu of clinical trials
Ultra-Rare Disease Approvals
New drug approval achieved with as few as 6 patient data points; 90% of FDA orphan drug approvals since 2014
Competitive Positioning
| Feature | Certara | Simulations Plus | Dassault Systemes |
|---|---|---|---|
| Core Positioning | End-to-end MIDD ecosystem | PBPK simulation & ADMET prediction | 3D modeling & molecular simulation |
| PBPK Platform | Simcyp (25-year history, 37-company consortium) | GastroPlus | BIOVIA |
| PK/PD Analysis | Phoenix WinNonlin (gold standard, 30-year history) | -- | -- |
| Regulatory Data Standards | Pinnacle 21 (used by FDA/PMDA) | -- | -- |
| AI Platform | Certara.AI (60M+ publications) | -- | -- |
| Market Access | BaseCase + HEOR consulting | -- | -- |
| Regulatory Agency Licenses | 11 agencies | Limited | Limited |
| Software + Services | Fully integrated | Primarily software | Primarily software |
Related Products
Start Your Model-Informed Drug Development Journey
From drug discovery to global regulatory approval and post-market, Certara's comprehensive software platform and consulting ecosystem can help you advance your science faster and with greater confidence
Contact Us to Learn More