Ennov Doc
Enterprise Document Management System (EDMS)
Metadata-Driven Unified Document Repository
Full Document Lifecycle Management
21 CFR Part 11 / EU Annex 11 Compliant
IDMP Integration Capability
Native AI Features (v11.0)
Cloud or On-Premise Deployment, Switchable Anytime
Ennov Doc is Ennov's enterprise document management software (EDMS), a comprehensive, flexible, scalable, and secure document management solution designed to help organizations streamline processes, improve operational efficiency, and eliminate regulatory compliance risks. As part of the broader Unified Compliance Platform, Ennov Doc consolidates dispersed documents and processes into a unified document repository, serving quality, regulatory, R&D, clinical, legal, commercial, and other departments.
Built on a metadata-driven document model, Ennov Doc provides the flexibility to adapt to any company's organizational needs. Ennov has over 25 years of experience, serves 450+ life science clients, has 500,000+ users globally, maintains a 98.5% on-time project delivery rate, and a 96% annual maintenance renewal rate.
Product Overview
Ennov Doc User Interface — document form with metadata fields (title, reference number, version, type)
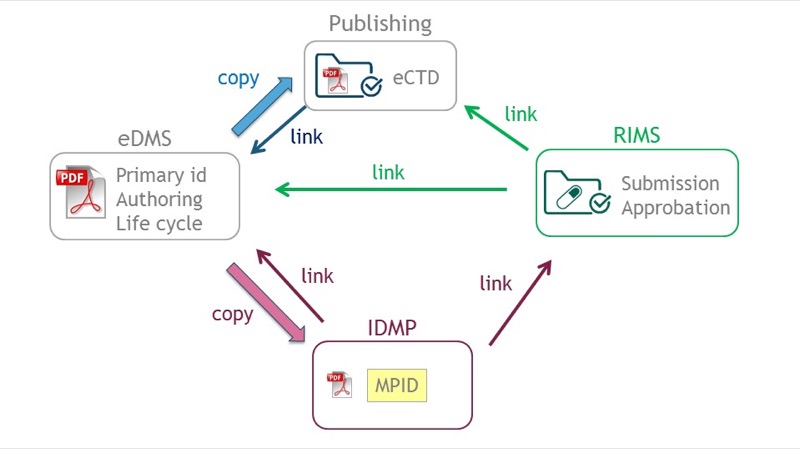
Cross-System Document Management — document linking and data flow across eDMS, publishing, RIMS, and IDMP
Unified Compliance Platform
Ennov Doc is part of a unified content and information management platform supporting the entire life science product development lifecycle. All modules share a single data repository — no inter-module integration required.
| Module | Domain | Description |
|---|---|---|
| Ennov Document | Document Management (EDMS) | Enterprise document management with full lifecycle, metadata, workflows, 21 CFR Part 11 compliance |
| Ennov Workflow | Business Process Management | Configurable workflow engine for quality events, CAPA, deviations, change control |
| Ennov Data | Data Management | Structured data management and regulatory data integration |
| Ennov Analytics | Data Visualization & Reporting | Dashboards, KPI reporting, Microsoft Power BI integration |
| Ennov AI | Artificial Intelligence | Native AI with LLM support, chatbot, automatic classification, MCQ generation, PV data extraction |
Core Features
Unified Access to All Documents
Consolidates dispersed documents and processes into a unified document repository. Metadata-driven document model, full-text and metadata search, integrated PDF viewer, DIA EDM Reference Model pre-configured
Document Lifecycle Management
Advanced lifecycle management (create, edit, review, approve, release), periodic review and expiration management, automatic PDF conversion, electronic signatures with meaning, version control and full revision history
Security & Compliance
21 CFR Part 11 and EU Annex 11 compliant, flexible permission management and role-based access control, LDAP/Active Directory synchronization, full audit trail, GxP compliance support
Enterprise Connectivity & Integration
MS Office 365, Google Drive, OneDrive connectivity. Open API / Web Services, DocShifter document conversion, Microsoft Power BI integration, cross-system document linking
AI Capabilities (Ennov AI — v11.0+)
Embedded chatbot, automatic MCQ generation from SOPs, eCTD document auto-classification, AI-driven agency correspondence extraction, EASI accelerates IDMP enrichment from 12 hours to 5 minutes
IDMP Integration Capability
Metadata-oriented architecture supporting IDMP controlled vocabularies, bidirectional data flow between eDMS and IDMP reference data, text mining support for extracting IDMP data from unstructured documents
IDMP Integration Architecture
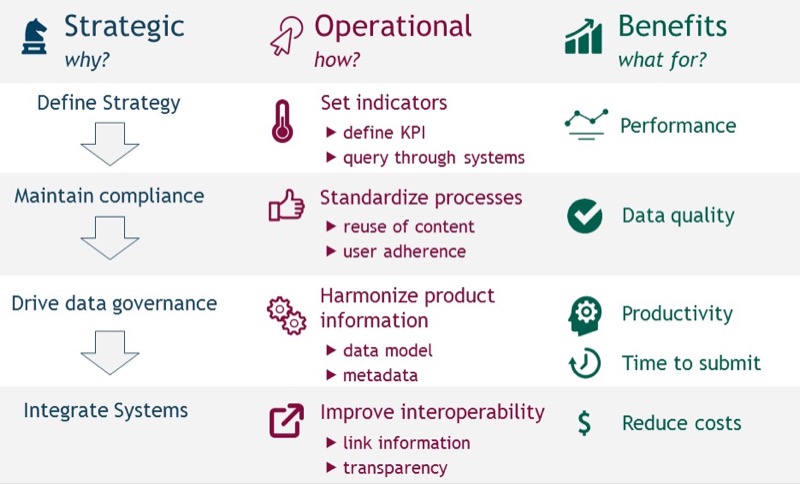
IDMP Governance Overview — strategy, operations, and benefits framework
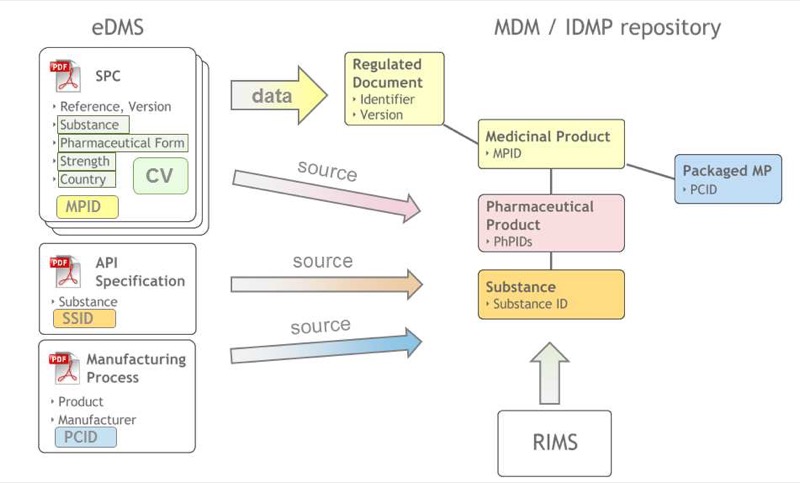
eDMS-IDMP Interaction — document and data flow architecture
Technical Specifications
| Specification | Details |
|---|---|
| Architecture | 100% web-based |
| Deployment | Cloud or on-premise (switchable at any time) |
| Cloud Hosting | Private cloud (Microsoft/OVH partners); single-tenant |
| Document Formats | Manages any document format; automatic PDF conversion |
| Search | Full-text and metadata-based search |
| Connectivity | MS Office 365, Google Drive, OneDrive |
| API | Open API / Web Services for custom integrations |
| AI | Native AI with LLM support (optional module, v11.0+) |
| Compliance | 21 CFR Part 11, EU Annex 11, GxP, GAMP 5, ISO 9001, ISO 27001 |
| Security | Flexible permission management, role-based access control, controlled print/copy |
| Administration | No-code configuration — no IT skills required |
Application Areas
Pharmaceutical
Quality document management, SOPs, regulatory submissions, GxP compliance, IDMP, eCTD publishing.
Biotechnology
R&D documents, regulatory submissions, clinical trial documents.
Medical Devices
Quality system documents, design controls, ISO 13485 compliance.
Contract Research Organizations (CRO)
Clinical operations documents, sponsor-specific document management.
Animal Health
Pharmacovigilance, regulatory compliance, quality documents.
Chemical / Specialty Chemicals
Quality documents, safety data sheets, environmental compliance (ISO 14001).
Competitive Comparison
| Feature | Ennov Doc | Veeva Vault QualityDocs | MasterControl | OpenText Documentum |
|---|---|---|---|---|
| Life Science Focus | Yes (core) | Yes (core) | Yes (core) | Yes (module) |
| On-Premise Option | Yes | No | Yes | Yes |
| Switchable Deployment | Yes (unique) | No | Limited | Limited |
| No-Code Configuration | Yes | Limited | Limited | No (requires IT) |
| eCTD Publishing | Yes (eCTD 4.0 certified) | Yes (Vault Submissions) | No | Limited |
| IDMP Integration | Yes (native + EASI) | Limited | No | Limited |
| AI Capabilities | Yes (native, v11.0) | Yes (Veeva AI) | Emerging | Limited |
| Pharmacovigilance | Yes (Ennov PV) | Limited | No | No |
Key Differentiators
Freedom to Choose Deployment
Cloud or on-premise deployment, switchable at any time — unique in the market
Unified Single Platform Architecture
All modules share one data repository — quality, regulatory, clinical, PV, training, and commercial, a scope few competitors can match
IDMP Ready
Native metadata architecture + EASI reduces manual enrichment work per product from 12 hours to just 5 minutes
25+ Years of Deep Experience
450+ clients, 500,000+ users, 98.5% on-time project delivery rate, 96% annual renewal rate, Calyx acquisition expanding to 1M+ global regulatory submissions
Customer Case Studies
| Customer | Industry | Key Outcomes |
|---|---|---|
| Pharmacosmos | Pharmaceutical (Denmark) | ~400 users across 7 countries; documents grew from 3,500 to 19,700; 50% CAPA cycle time reduction |
| Horiba | Scientific Instruments (Japan/Global) | Standardized workflows; eliminated paper; reduced search time; improved cross-department collaboration |
| Seqens | Pharmaceutical Synthesis (CDMO) | Global document management and quality platform; FDA/ANSM/EMA compliance; data integrity |
| HTL Biotechnology | Biotechnology | Eliminated signature bottlenecks; saved ~2 FTEs; prepared for global expansion |
Related Products
Implement Ennov Doc Enterprise Document Management
Simplify document management through a unified document repository, ensure regulatory compliance, and support the complete document lifecycle from quality to regulatory
Contact Us