Ennov QMS
Unified Compliance Platform Quality Suite — Integrating Documents, Quality, Training, and Analytics, Trusted by 450+ Life Science Clients
Ennov QMS is a comprehensive, fully integrated electronic Quality Management System (eQMS) primarily designed for regulated life science industries — pharmaceutical, biotechnology, medical devices, animal health, cosmetics, and healthcare. It is part of the broader Ennov Quality Suite, which is built on the Unified Compliance Platform — a single platform that also supports regulatory affairs, clinical trials, pharmacovigilance, and commercial compliance.
Ennov QMS manages quality processes (deviations, CAPAs, change control, audits, complaints, etc.) through configurable workflows, with a visual form builder and graphical workflow modeling tool — all configuration can be done by functional administrators without programming or IT skills. Ennov positions itself as "the only GxP platform with AI natively embedded in day-to-day operations," with 450+ life science clients and 500,000+ users.

Next-Generation eQMS Key Challenges — system integration, compliance management, data insights, and scalability
Quality Suite Composition
The Quality Suite combines four core modules on a single unified platform, sharing a single database and single user interface, eliminating the need for inter-module system integration:
Ennov Doc for Quality
Comprehensive GxP document management, sharing, and tracking. Pre-configured to align with the DIA EDM Reference Model, supporting full document lifecycle management, 21 CFR Part 11 compliant.
Ennov QMS
Manages quality events/processes through configurable workflows. Visual form builder, graphical workflow modeling tool, smart data forms, and real-time monitoring dashboards.
Ennov Training
Fully integrated electronic Learning Management System (eLMS) designed for GxP training compliance. Document version updates automatically trigger training workflows, with AI-driven MCQ auto-generation.
Ennov Analytics
Native data visualization with pre-configured dashboards based on industry sources. Integrates with Microsoft Power BI, focusing on risk management, progress tracking, and compliance metrics.
Quality Event Management
| Quality Event Type | Description |
|---|---|
| CAPAs | Complete CAPA lifecycle management (Corrective and Preventive Actions) |
| Deviations | Recording, tracking, and resolution of deviations |
| Non-Conformances | Non-conforming product/process management |
| Complaints | Automated recording, tracking, and resolution |
| Change Control | Change management workflows |
| Audits | Comprehensive management of audit plans, individual audits, and findings |
| OOS Investigations | Pre-defined processes for out-of-specification handling |
| Supplier Quality | Supplier quality management processes |
| Risk Management | Quality risk management processes |
| Design Controls | Design control processes (for medical devices) |
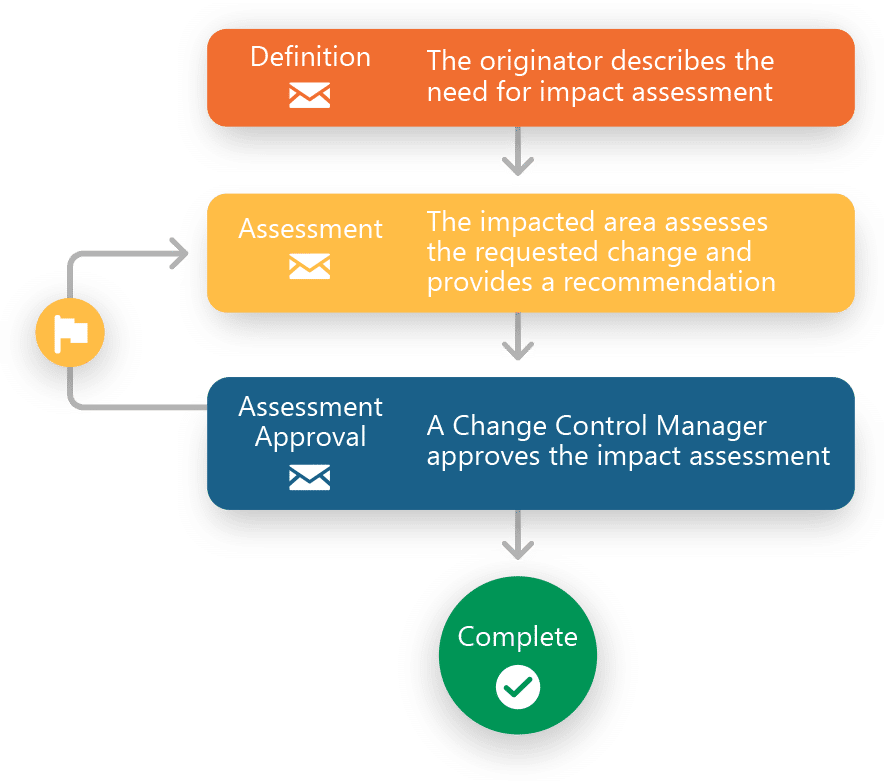
Ennov QMS Change Control Process — visual workflow management from definition, impact assessment, approval to completion
Core Features
Configurable Workflows
Visual graphical workflow builder requiring no IT/programming skills. Smart data forms with conditional branching and metadata rules, workflow validation with built-in consistency checks
AI Capabilities (Ennov AI — v11.0+)
Training MCQ auto-generation, AI chatbot assistant, predictive analytics, automatic classification. All AI operations are logged, validated, fully auditable, and natively embedded in the platform
Pre-Configured Reference Models
DIA EDM Reference Model alignment, Ennov Quality Core Model providing pre-defined processes and workflows based on industry best practices, accelerating deployment
Unified Platform Architecture
Doc, QMS, Training, and Analytics share a single database and UI — no integration complexity. Document version updates automatically trigger training, quality events directly link to training records
Future-Proofing Quality Management White Paper — next-generation eQMS adoption guide for life science SMEs
Technical Specifications
| Specification | Details |
|---|---|
| Architecture | 100% web-based; Unified Compliance Platform |
| Deployment | Cloud (SaaS), on-premise, or hybrid — switchable at any time |
| Database | Single shared database across all modules |
| Configuration | No-code — visual form builder and graphical workflow designer |
| Compliance | 21 CFR Part 11, EU Annex 11, ISO 9001, ISO 13485, ISO 14971, GxP |
| Certifications (Company) | ISO/IEC 27001, ISO 9001:2015 |
| Integration | REST API open to any programming environment/application platform |
| Analytics | Native Ennov Analytics; Microsoft Power BI integration |
| AI | Ennov AI (optional module, v11.0+) — MCQ generation, chatbot, predictive analytics |
| Security | Role-based access control, electronic signatures, full audit trail |
| Multilingual | English, French, German, Japanese, Simplified Chinese, and 13+ languages |
Application Areas
Pharmaceutical
GMP compliance, CAPAs, deviations, change control, SOPs, audits, QbD.
Biotechnology
Quality documentation, regulatory compliance, process control.
Medical Devices
Design controls, ISO 13485 compliance, risk management (ISO 14971).
Animal Health
Quality documentation, pharmacovigilance, regulatory compliance.
Contract Development & Manufacturing (CDMO)
GMP documentation, supplier quality, batch records.
Cosmetics
Quality management, GMP compliance, document control.

Ennov QMS Intelligent Quality Management — AI-driven task tracking, document parsing, and training management interface
Future-Ready eQMS Key Attributes
Seamless Connectivity
Real-time communication for all stakeholders; automated compliance tasks to reduce workload on lean teams
Guaranteed Accountability
Rigorous tracking of all discrepancies and changes to resolution; clear audit trails and change history
Integrated Systems
Linking data, documents, and processes; "one-stop" integration of multiple functions
Proactive Compliance
Automated compliance updates and alert systems; real-time adaptation to regulatory changes
Collaborative Solutions
Tools for effective problem-solving and information sharing; leveraging collective expertise
Operational Transparency
Comprehensive dashboards, detailed reports, and clear visualizations; risk management visibility and informed decision-making
Competitive Comparison
| Feature | Ennov Quality Suite | Veeva Vault QMS | MasterControl | ETQ Reliance |
|---|---|---|---|---|
| Target | Life sciences (all sizes) | Enterprise life sciences | Life sciences, manufacturing | Manufacturing, life sciences |
| Deployment | Cloud, on-premise, hybrid | Cloud only (SaaS) | Cloud, on-premise | Cloud, on-premise |
| Integrated EDMS | Yes (native) | Yes (Vault) | Yes | Yes |
| Integrated Training | Yes (native eLMS) | Yes | Yes | Add-on |
| AI Capabilities | Native (v11.0) | Yes (Veeva AI) | Emerging | Yes |
| No-Code Configuration | Yes | Limited | Limited | Yes (no-code) |
| Pre-Configured Models | DIA GMP + Quality Core | No | Limited | No |
| Switchable Deployment | Yes (unique) | No | Limited | Limited |
Key Differentiators
Unified Platform Architecture
Doc, QMS, Training, and Analytics share a single database and UI — no integration complexity
Native AI
Claims to be the only GxP platform with AI natively embedded in daily operations (v11.0+), including chatbot, MCQ auto-generation, and predictive analytics
Deployment Flexibility
Cloud, on-premise, or hybrid — switchable at any time, a unique flexibility in the market
Proven ROI
50% CAPA cycle time reduction (Pharmacosmos), ~2 FTE savings (HTL Biotechnology), 98.5% on-time project delivery rate

Ennov QMS Document Management — role-based access control (super user/administrator/consumer) and audit trail
Customer Case Studies
| Customer | Industry | Key Outcomes |
|---|---|---|
| Pharmacosmos | Pharmaceutical (Denmark) | 400 users across 7 countries; 50% CAPA cycle time reduction; paperless operations |
| HTL Biotechnology | Biotechnology | Eliminated signature bottlenecks; saved ~2 FTEs; laid foundation for global expansion |
| Vetoquinol | Animal Health | Centralized QMS eliminated data silos across multiple sites |
| Septodont | Dental Pharmaceutical | Quality and regulatory document/process management |
Related Products
Implement the Ennov QMS Quality Suite
Simplify quality management and accelerate compliance through a unified platform, transforming from reactive to predictive quality management
Contact Us